Every time a doctor checks your blood pressure digitally, every time a hospital monitor beeps to signal your heart rate, and every time a surgeon uses a robotic-assisted tool, there is an invisible world of electronic components making it all possible. These components, ranging from tiny transistors to large display systems, are the backbone of modern healthcare technology.
For patients and caregivers, understanding how medical devices work and why their reliability matters is more important than ever. We live in an era where your health literally depends on the quality of the hardware running behind the scenes.
1. What Are Electronic Components in Medical Devices?
Medical devices are not just mechanical tools. Sophisticated electronic systems power most modern health equipment. At the core of these systems are electronic components, small but critical parts that control, process, and transmit information within the device.
Common electronic components found in medical devices include:
- Microprocessors and chips: The brain of any digital health device, processing data and executing instructions.
- Transistors: Tiny semiconductor switches that amplify electrical signals or toggle circuits on and off, enabling everything from hearing aids to MRI machines to function precisely.
- Sensors: Detect physical or chemical changes such as blood glucose levels, oxygen saturation, or body temperature.
- Capacitors and resistors: Regulate and stabilize electrical flow, preventing damage to both the device and the patient.
- Display panels: Show readings, waveforms, and imaging data to clinicians and patients.
Without these parts working in harmony, the device cannot function safely or accurately.
Why Component Quality Directly Affects Patient Safety
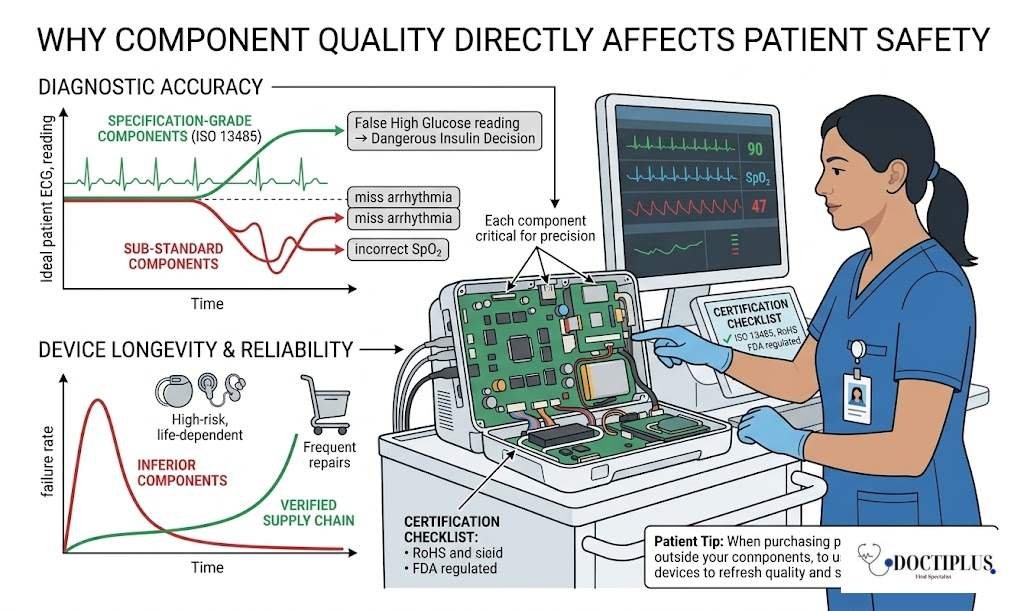
Does it really matter where the parts inside a medical device come from? The answer is unequivocally yes. Component quality is one of the most overlooked factors in patient safety.
Accuracy of Diagnostics
Diagnostic equipment such as blood glucose monitors, ECG machines, and pulse oximeters relies on sensors and transistors that must deliver highly accurate readings. A malfunctioning component can lead to:
- False high or false low glucose readings in diabetic patients can potentially trigger dangerous insulin decisions.
- Inaccurate ECG outputs could miss life-threatening arrhythmias.
- Incorrect oxygen saturation readings are especially critical in the ICU and surgical settings.
Device Longevity and Reliability
Medical devices are expected to perform without failure for years, sometimes decades. When inferior components are used, devices break down more frequently, require costly repairs, and may fail at the worst possible moment. For patients who depend on implantable devices like pacemakers or cochlear implants, this is not just inconvenient; it can be life-threatening.
Role of Trusted Supply Chains
Healthcare manufacturers source their components from verified, regulated suppliers. For example, reputable electronic transistor wholesale distributors must comply with international standards such as ISO 13485 (quality management for medical devices), RoHS (restriction of hazardous substances), and FDA regulatory requirements. These certifications ensure that every transistor, chip, or sensor meets strict benchmarks before it ever reaches a hospital device.
Patient Tip:
When purchasing personal health devices such as glucose monitors or digital blood pressure cuffs, always look for products bearing CE, FDA clearance, or ISO certification marks. These indicate that the device’s components and manufacturing process have been independently verified.
Medical Devices That Depend Most on Electronic Components
Wearable Health Monitors
Wearables like smartwatches with health tracking, continuous glucose monitors (CGMs), and ECG patches have transformed how patients manage chronic conditions. These devices are packed with miniaturized sensors and transistors that must maintain accuracy 24 hours a day.
Key components in wearables include:
- Optical sensors for heart rate and SpO2 monitoring.
- Accelerometers to detect movement and sleep patterns.
- Low-power Bluetooth chips for data transmission to smartphones.
- Bioimpedance sensors for body composition analysis.
The reliability of these components determines whether a wearable gives you trustworthy data or dangerously misleading readings.
Hospital Diagnostic Equipment
Hospitals use a broad range of diagnostic tools that depend entirely on precision electronics:
- MRI and CT scanners use thousands of electronic components to generate and process imaging data.
- Ventilators depend on sensors and microcontrollers to deliver the exact volume and pressure of air a patient needs.
- Infusion pumps use precision electronic flow controllers to administer medications at exact doses.
- Patient monitoring systems track multiple vital signs simultaneously across hospital wards.
A single faulty component in any of these systems can delay diagnosis, reduce treatment accuracy, or, in critical cases, endanger life.
Telehealth and Remote Patient Monitoring
Telehealth has expanded rapidly, particularly since the COVID-19 pandemic. Remote monitoring devices now allow patients with chronic conditions such as heart disease, diabetes, or COPD to be observed from their homes. These systems rely on:
- High-quality transmission chips for stable video and data connections.
- Medical-grade display technology for accurate visual consultations.
- Sensors that replicate clinical-level accuracy in a home environment.
The effectiveness of telehealth is only as good as the hardware enabling it. Poor-quality components lead to dropped connections, inaccurate readings, and a reduced quality of remote care.
Hospital Display Systems and Telemedicine Platforms
Modern hospitals and clinics increasingly rely on advanced collaborative display systems for medical education, case review, and interdisciplinary consultations. Solutions provided by all-in-one interactive meeting boards manufacturers are now found in medical school training rooms, surgical briefing suites, and telemedicine hubs. These boards allow multiple clinicians to simultaneously review patient imaging, annotate findings, and coordinate treatment plans in real time, significantly reducing communication errors that contribute to adverse patient outcomes.
4. Regulatory Standards That Protect Patients
To protect patients from the risks of substandard components, multiple international and national bodies enforce strict regulations on medical device manufacturing.
FDA (United States)
The U.S. Food and Drug Administration requires that all medical devices sold in the United States meet rigorous safety and efficacy standards. Medical device manufacturers must submit evidence that their products, including the electronic components within them, perform safely under real-world conditions.
CE Marking (European Union)
In the EU, medical devices must carry the CE mark, confirming compliance with the EU Medical Device Regulation (MDR). This includes requirements for the electronic components and software within the device.
ISO 13485
This international standard specifically governs quality management systems for medical device manufacturers. It ensures systematic approaches to design, development, and post-market surveillance, covering the entire supply chain, including electronic component sourcing.
RoHS Compliance
The Restriction of Hazardous Substances directive limits the use of dangerous materials in electronics. For medical devices, this is critical because patients or healthcare workers may have prolonged contact with these devices.
What Patients Should Know:
Before using any medical device, particularly devices purchased online or from non-regulated markets, check for official certifications. Counterfeit or uncertified medical devices may contain substandard components that deliver inaccurate results or cause electrical hazards.
Red Flags: Signs a Medical Device May Have Component Issues
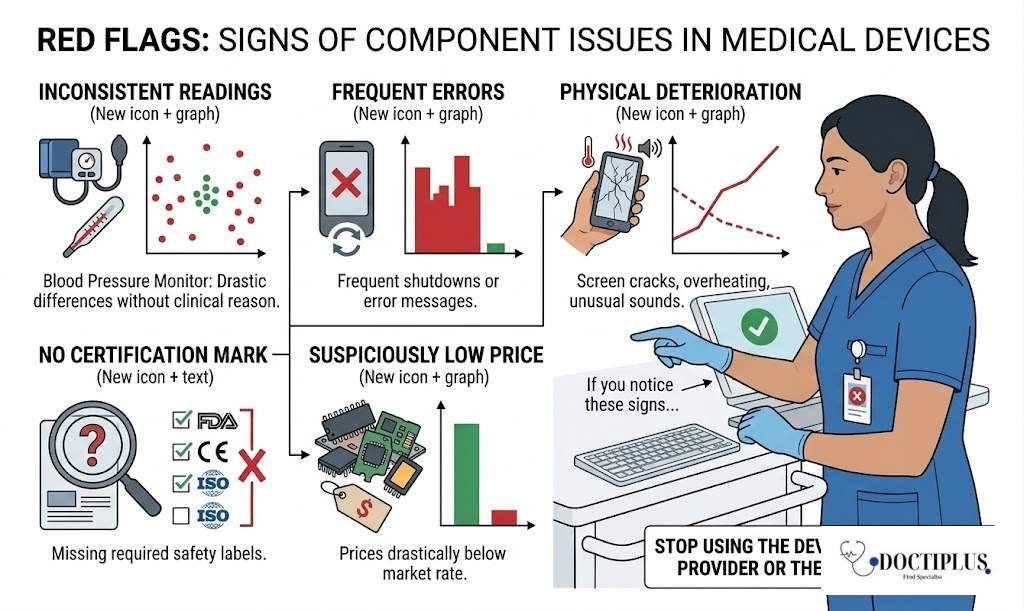
Patients and caregivers should be aware of warning signs that a medical device may not be functioning reliably due to component problems:
- Inconsistent readings: A blood pressure monitor that gives very different results in rapid succession without a clear clinical reason.
- Frequent error messages or shutdowns: A device that frequently restarts or displays error codes unprompted.
- Physical deterioration: Cracked screens, overheating during normal use, or unusual sounds from electronic components.
- No certification mark: A device without FDA, CE, or ISO markings purchased from an unknown source.
- Very low price from unverified sellers: Prices drastically below market rate may indicate counterfeit parts.
If you notice any of these signs, stop using the device and consult your healthcare provider or the device manufacturer.
Practical Advice for Patients and Healthcare Providers
For Patients
- Always purchase medical devices from licensed pharmacies, hospitals, or verified medical supply retailers.
- Register your device with the manufacturer so you receive safety alerts and recalls.
- Follow manufacturer maintenance guidelines, including battery replacement and software updates.
- Do not attempt to repair electronic medical devices yourself, as this can damage sensitive components.
- Report malfunctions to the manufacturer and, if relevant, to your national regulatory authority, such as the FDA MedWatch program.
For Healthcare Professionals and Administrators
- Source medical devices only from manufacturers who demonstrate full supply chain transparency and component traceability.
- Require documentation of component certifications when procuring new equipment.
- Establish a preventive maintenance program for electronic medical equipment.
- Train staff to recognize early signs of equipment malfunction due to component degradation.
- Stay updated on device recalls issued by the FDA, MHRA, or relevant national body.
Future of Health Technology and Electronic Components
The relationship between healthcare and electronics is growing more complex and more critical. Emerging technologies such as AI-powered diagnostics, implantable biosensors, robotic surgery, and personalized medicine all rely on next-generation electronic components that are smaller, faster, and more powerful than ever before.
As these technologies develop, the standards governing the components inside them must evolve too. Patients can expect:
- More precise wearable sensors capable of detecting disease biomarkers in real time.
- AI-integrated diagnostic tools that reduce human error in imaging interpretation.
- Implantable devices with decade-long battery lives powered by energy-harvesting components.
- Smart hospital environments where electronic systems coordinate patient care automatically.
Each of these innovations depends on advances in electronic component technology and on the regulatory frameworks that ensure those components meet medical-grade standards.
Conclusion
Electronic components are the silent foundation of modern healthcare. From the transistor in your glucose monitor to the display system in your doctor’s consultation room, these parts determine whether your care is safe, accurate, and effective.
As a patient, being informed about the technology behind your medical devices empowers you to make better decisions, ask better questions, and recognize when something might not be right. As a healthcare professional or administrator, this reinforces why component quality and supplier accountability are not just technical concerns but patient safety concerns.
When in doubt, always consult a qualified medical professional and choose certified, regulated medical devices from trusted sources.
References and Further Reading
- U.S. Food and Drug Administration. Medical Devices Overview [Accessed 2025]
- International Organization for Standardization. ISO 13485: Medical devices quality management systems [Accessed 2025]
- European Commission. EU Medical Device Regulation (MDR) 2017/745 [Accessed 2025]
- World Health Organization. Medical devices: managing the mismatch [Accessed 2025]
- National Institute of Biomedical Imaging and Bioengineering. Wearable Biosensors [Accessed 2025]
- RoHS Guide. RoHS Compliance for Medical Electronics [Accessed 2025]
- U.S. FDA MedWatch. Safety Reporting Portal [Accessed 2025]
Medical Disclaimer
This article has been prepared for general informational and educational purposes only. Doctiplus and its contributors do not provide medical advice. The content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or the use of a medical device. Never disregard professional medical advice or delay seeking it because of something you have read in this article.

