The rapid growth of digital-first weight loss clinics in the UK has generated a question that matters clinically: do these services prescribe and monitor safely, or does the removal of face-to-face interaction compromise patient outcomes? The answer, as with most things in medicine, depends entirely on which clinic you are asking about.
Published safety data from a 125,000-member supervised UK cohort offers the most granular answer currently available. This data comes from Voy, a CQC-regulated digital-first provider prescribing tirzepatide and semaglutide, with outcomes published in peer-reviewed journals including Diabetes, Obesity and Metabolism and JMIR. This article examines what that data shows, what the regulator has found across the broader sector, and what the evidence tells us about digital delivery as a model for GLP-1 weight loss treatment.
Published Safety Data: What 125,000 Patients Show
The headline finding from the Voy outcome data, published in Diabetes, Obesity and Metabolism in 2025, is a 96% relative reduction in prescribing errors compared to external benchmarks. The Voy prescribing error rate was 0.17%, against an external comparator rate of 4.43%. In practical terms, this means that for every 100 prescriptions issued externally with an error, fewer than four occurred within the Voy model.
Prescribing errors in GLP-1 weight loss treatment carry specific risks. The dose-escalation schedule for tirzepatide runs from 2.5mg to a maximum of 15mg over 20 weeks. An incorrect starting dose, an escalation that skips a step, or a dose issued without an appropriate clinical context can produce significant gastrointestinal adverse events. Real-world FDA adverse event data show that “incorrect dose administered” was the most common adverse event signal for tirzepatide between 2022 and 2025, accounting for 19,461 reports across that period. The dose-escalation complexity is precisely where prescribing governance matters most, and the 96% relative error reduction demonstrates what structured digital prescribing can achieve.
96% relative reduction in prescribing errors: 0.17% in the supervised Voy cohort versus 4.43% in external benchmarks. “Incorrect dose administered” was the most commonly reported adverse event signal for tirzepatide in FDA pharmacovigilance data 2022-2025.
Overall Safety-Incident Rate: 0.16%
The overall safety-incident rate in the cohort was 0.16%. For a cohort of 125,000 members, that represents a very small absolute number of safety incidents across a complex clinical population managing a chronic condition with a prescription medication that requires ongoing dose management.
For context, clinical trials of tirzepatide reported serious adverse events in 4.3% to 7.1% of participants across different dose groups. However, those figures capture all serious events over 72 weeks regardless of their relationship to the medication. A 0.16% safety-incident rate in a real-world clinical population, where the patient profile is more heterogeneous than in a controlled trial, reflects robust monitoring and proactive intervention.
Side Effect Reduction: 43% Below External Benchmarks
Perhaps the most clinically meaningful figure is the 43% reduction in side effects compared to external benchmarks. Side effects are the primary driver of treatment discontinuation, and discontinuation is the primary predictor of poor long-term weight loss outcomes. A cohort that experiences materially fewer side effects is, by extension, a cohort that stays on treatment longer and achieves better outcomes.
The mechanism for this reduction is dose management. Published data from the same cohort shows side effects declining steeply over time: nausea from 35% to 8%, constipation from 33% to 10%, and headaches from 28% to 5% over 12 months (published in Cureus, 2025). This trajectory reflects supervised prescribing that adjusts escalation pace to match individual tolerance rather than advancing on a fixed schedule regardless of how the patient is responding.
For eligible patients, Voy provides CQC-regulated prescribing with the clinical governance model described above.
What The Regulator Says About The Broader Sector

The General Pharmaceutical Council published its themed review of weight management medicines and services in April 2026, drawing on inspection data and concerns received between January 2024 and December 2025. The review examined 77 pharmacies providing weight management services and found 106 individual standards failures across those businesses.
The most commonly identified failures were:
- Absence of independent BMI verification. From February 2025, the GPhC requires online pharmacies to independently verify weight, height, and BMI rather than relying on self-reported data. Many providers were still not meeting this standard.
- Incomplete patient consultation records and inadequate documentation of clinical decision-making.
- Weaknesses in risk assessment procedures and clinical governance arrangements.
- Prescribing to individuals with a history of eating disorders without appropriate screening or referral pathways.
- No ongoing clinical follow-up after initial prescribing.
Online pharmacies accounted for 49% of concerns raised with the regulator, compared to 26% for community pharmacies. The regulator was explicit: standards are “not always consistently met” across the sector. This is the regulatory environment within which the Voy safety data of 0.16% safety incidents and a 96% prescribing error reduction stands out.
GPhC themed review: 106 individual standards failures identified across 77 pharmacies providing weight management services, between January 2024 and December 2025. Online pharmacies accounted for 49% of all concerns raised. Standards were “not always consistently met.”
BBC Investigation: Falsification of Clinical Information
In June 2025, an undercover BBC investigation found that two online pharmacies permitted patients to falsify health information to obtain access to GLP-1 medications, despite regulatory rules specifically designed to prevent this. This is the safety failure that independent BMI verification and structured clinical assessment are designed to prevent. The investigation prompted further regulatory scrutiny and reinforced the GPhC’s position that assessment quality cannot depend on patient self-report alone.
The Pharmaceutical Journal noted that the surging demand for GLP-1s has created a two-tier market: on one side, regulated providers with robust clinical assessment; on the other, services that may prescribe without meaningful governance. The clinical risk profile of these two tiers is not equivalent.
What the Evidence Shows About Digital GLP-1 Delivery
JMIR 2025: Remote Delivery Achieves Trial-Comparable Outcomes
A 2025 study published in JMIR Formative Research evaluated 339 participants completing a 12-month remote GLP-1 weight management cohort, combining medication, app-based behavioural support, coaching from registered dietitians, and clinical oversight. Both the tirzepatide and semaglutide cohorts exceeded clinically significant weight loss thresholds with acceptable safety profiles.
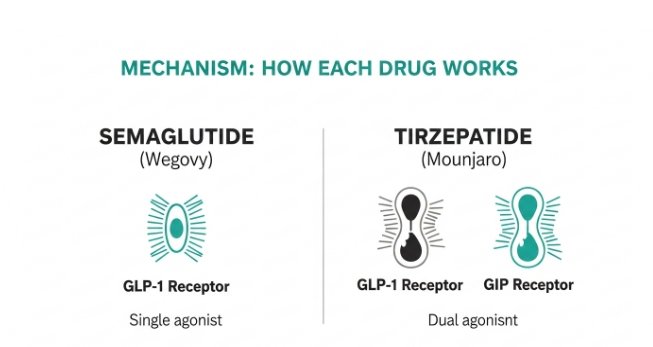
The study concluded that remotely delivered GLP-1 weight management can achieve outcomes that align closely with clinical trial results, and may reduce healthcare costs by 10% to 70% compared to traditional UK services. This supports the clinical case that the delivery modality, digital versus in-person, is not the determinant of safety or efficacy. The governance model is.
JMIR 2025: Voy Real-World Data Exceeds Trial Benchmarks
The same JMIR publication that reported 100% quality of life improvement also recorded 24% average weight loss at 12 months across a cohort of 125,000+ members, compared to 16% in clinical trials of the same medication. This 50% improvement over trial benchmarks is itself a safety-relevant finding: it reflects greater treatment continuity, more effective dose management, and better side effect control than the trial population achieved.
The 35% relative reduction in treatment discontinuation published from the same cohort is the underlying mechanism. Members who stay on treatment produce better outcomes. Members who experience fewer side effects stay on treatment. The 43% side effect reduction and the 35% discontinuation reduction are causally connected outcomes of the same governance model.
24% average weight loss at 12 months versus 16% in clinical trials of the same medication. 35% relative reduction in treatment discontinuation. 100% quality of life improvement. Published in JMIR, 2025 (N=125,000+).
Regulatory Framework: What Safe Digital Prescribing Requires
GPhC Standards For Online Pharmacies
All pharmacies in Great Britain, including online pharmacies, must be registered with the General Pharmaceutical Council and meet its standards. Since February 2025, online pharmacies have been required to independently verify weight and BMI rather than relying on self-reported data. This is a minimum requirement, not a quality differentiator. Pharmacies that are not meeting it are operating below the regulatory floor.
The GPhC’s April 2026 review sets out explicit standards for compliant weight management services:
- Robust, regularly reviewed risk assessments and standard operating procedures.
- Effective clinical governance and oversight, including appropriate due diligence when working with third-party prescribing services.
- Independent verification of clinical information, including weight and BMI.
- Individual review of clinical suitability, with specific consideration of eating disorder risk.
- All consultations and clinical decision-making to be thoroughly documented.
- Clear communication and accessible ongoing support.
CQC Regulation And What it Adds
The Care Quality Commission regulates healthcare providers in England, including digital-first clinics that fall within its scope. CQC registration requires demonstrated standards of safe care, effective care, responsive care, caring, and well-led governance. Not all online pharmacies prescribing GLP-1 medications are CQC-registered; those that are have undergone a layer of regulatory scrutiny that pharmacy registration alone does not provide.

The MHRA regulates the medicines themselves: all licensed GLP-1 medications in the UK carry a UK marketing authorisation (identifiable by a PL number on the packaging). CQC regulates the care delivery model. GPhC regulates the pharmacy dispensing the medication. A provider operating across all three layers of regulation is operating in a materially different risk environment from one operating under pharmacy registration alone.
How To Evaluate A Digital Weight Loss Clinic
The published data establishes what best-in-class digital-first prescribing looks like. The regulatory reviews establish where the sector falls short. The practical question for clinicians and patients is how to distinguish between them.
Questions That Dstinguish Regulated From Unregulated Access
- Is the pharmacy GPhC-registered? All UK pharmacies must be. Check at pharmacyregulation.org. The GPhC register is publicly searchable.
- Is BMI independently verified? Since February 2025, this has been a regulatory requirement. Any provider that does not independently verify weight and BMI is not meeting minimum GPhC standards.
- Is there a qualified prescriber assessment? GLP-1 medications are prescription-only. They must be prescribed by a GMC-registered prescriber following individual clinical assessment, not issued via questionnaire alone.
- Does the provider offer ongoing monitoring? Dose escalation for tirzepatide spans 20 weeks. A provider that prescribes without follow-up is not managing the escalation period, where most adverse events occur.
- Does the provider publish safety and outcome data? Transparency about safety incident rates, prescribing error rates, and patient outcomes is a meaningful differentiator. Providers confident in their governance publish it.
- Is the provider CQC-regulated? Not a universal requirement for all pharmacy models, but a significant additional governance layer for digital-first clinic models.
Less than half of UK adults are aware that GLP-1 weight loss injections are prescription-only medicines. Between February 2025 and January 2026, the ASA identified approximately 900 advertisements from 38 companies likely to break rules on promoting prescription medicines to the public.
FAQs
| Question | Answer |
|---|---|
| Are digital weight loss clinics safe? | It depends entirely on the specific clinic and its governance model. Published safety data from the best-regulated digital-first providers show outcomes that match or exceed in-person care benchmarks. A 125,000-member UK cohort published in Diabetes Obesity and Metabolism in 2025 showed a 0.16% overall safety-incident rate and a 96% relative reduction in prescribing errors versus external benchmarks. The GPhC’s April 2026 review of the broader sector found 106 standards failures across 77 pharmacies inspected. Both findings are simultaneously true: excellent governance produces excellent outcomes, and inadequate governance produces preventable failures. |
| What does the GPhC regulation require for weight loss clinics? | Since February 2025, GPhC-registered pharmacies have been required to independently verify weight, height, and BMI rather than relying on patient self-report. They must carry out individual clinical assessments before prescribing, maintain complete consultation records, conduct appropriate risk assessments, screen for eating disorders, and provide ongoing monitoring throughout treatment. The GPhC’s April 2026 themed review found these standards were not consistently met across the sector. |
| What is the prescribing error rate for GLP-1 medications? | Across external benchmarks, the prescribing error rate for GLP-1 medications is 4.43%, according to published comparator data. In a supervised UK cohort of 125,000+ members, the prescribing error rate was 0.17%, representing a 96% relative reduction. FDA pharmacovigilance data from 2022 to 2025 show that “incorrect dose administered” was the single most commonly reported adverse event signal for tirzepatide, highlighting why dose accuracy is the most clinically important dimension of GLP-1 prescribing quality. |
| Can digital weight loss clinics match clinical trial outcomes? | In well-governed models, yes. A JMIR 2025 study of 339 participants in a remote GLP-1 weight management cohort found outcomes that aligned closely with clinical trial results. The Voy JMIR 2025 publication of 125,000+ members found 24% average weight loss at 12 months, compared to 16% in clinical trials of the same medication. The difference between digital clinics that match or exceed trial outcomes and those that fall short reflects governance quality, not delivery channel. |
| What should |
This article is for informational purposes and does not constitute medical advice. Always consult a qualified healthcare professional before starting any weight loss treatment. Individual results may vary. Treatment is subject to clinical suitability assessment.
Sources
- • Voy outcome data. Published in Diabetes, Obesity and Metabolism, 2025. 96% relative prescribing error reduction; 0.17% vs 4.43% external benchmark; 0.16% safety-incident rate; 43% side effect reduction. N=125,000+.
- • Voy outcome data. Published in JMIR, 2025 (N=125,000+). 24% average weight loss at 12 months; 100% quality of life improvement; 35% relative reduction in treatment discontinuation.
- • Voy outcome data. Published in Cureus, 2025. Side effect trajectory: nausea 35% to 8%, constipation 33% to 10%, and headaches 28% to 5% over 12 months.
- • GPhC. Weight management medicines and services: A review of GPhC inspections and concerns. April 2026. https://www.pharmacyregulation.org/about-us/news-and-updates/gphc-calls-pharmacies-strengthen-safeguards-and-governance-weight-management-services
- • MHRA. Warns of unsafe fake weight loss pens. GOV.UK, 2023. https://www.gov.uk/government/news/mhra-warns-of-unsafe-fake-weight-loss-pens
- • MHRA. Urges the public to avoid illegal online weight-loss medicines. GOV.UK, December 2025. https://www.gov.uk/government/news/mhra-urges-public-to-avoid-illegal-online-weight-loss-medicines-this-new-year
- • GPhC. New rules for online pharmacies: independent BMI verification required. February 2025. https://www.pharmacyregulation.org
- Richards R et al. Semaglutide and Tirzepatide in a Remote Weight Management Program: 12-Month Retrospective Observational Study. JMIR Formative Research, 2025. N=339. https://formative.jmir.org/2025/1/e81912
- Pharmaceutical Journal. Everything you need to know about GLP-1s for weight loss. February 2026. (GPhC-cited MHRA Yellow Card data, 2020-2025.)
- Pharmaceutical Journal. The dark side of the miracle jab: why eating disorder safety cannot be an afterthought. February 2026.
- PMC. Real-world safety concerns of tirzepatide: FAERS retrospective analysis 2022-2025. (Incorrect dose administered: 19,461 reports.)
- Pharmacy Magazine. Less than half of UK adults are aware that GLP-1s are prescription medicines. April 2026.
- Jastreboff AM et al. SURMOUNT-1. New England Journal of Medicine, 2022. N=2,539.