What if your own immune system could be re-engineered to hunt down and destroy cancer precisely, powerfully, and permanently? That’s not a sci-fi fantasy anymore. It’s happening right now in hospitals around the world.
What Is CAR-T Cell Therapy? Let’s Start from the BeginningA Brief but Remarkable HistoryHow Exactly Does It Work? The Step-by-Step ProcessWhat Cancers Can It Treat? Current Approvals and ResultsFDA-Approved CAR-T Therapies (as of 2026)Challenge: Solid Tumours Are a Different BeastWhat Are the Side Effects? Being Honest About the Risks1. Cytokine Release Syndrome (CRS)2. ICANS (Immune Effector Cell-Associated Neurotoxicity Syndrome)Next Frontier: What’s Coming in CAR-T Science“Off-the-Shelf” CAR-T CellsMulti-Targeted CAR-T CellsCRISPR-Engineered CAR-T CellsCAR-T Beyond CancerWhy This Matters: The Bigger PictureRole of a CAR-T Specialist: Who Guides You Through This Journey?Choosing the Right CAR-T Cell Therapy HospitalFinal ConclusionMedical DisclaimerReferences
What Is CAR-T Cell Therapy? Let’s Start from the Beginning
Cancer has always been medicine’s most stubborn enemy. For decades, the go-to weapons were chemotherapy, radiation, and surgery treatments that work, but often at a brutal cost to the patient’s body. Then came a revolution: immunotherapy, the idea that the human immune system itself could be trained to fight cancer.
CAR-T cell therapy is one of the most exciting and powerful branches of immunotherapy today. CAR stands for Chimeric Antigen Receptor, a mouthful, yes, but the concept is surprisingly elegant.
Here’s the big idea in plain language:
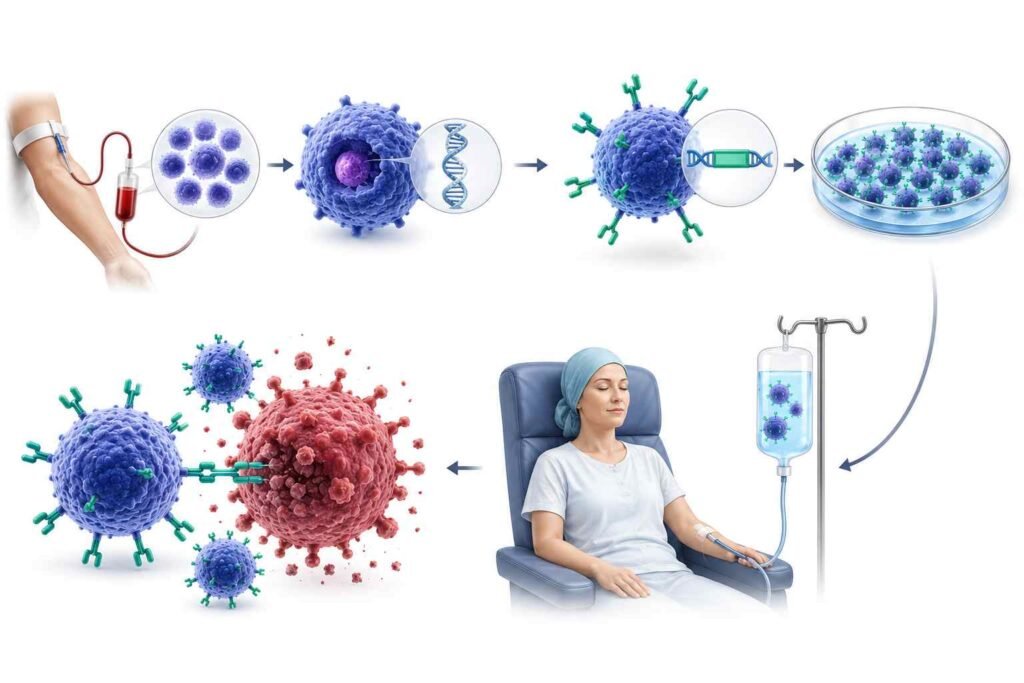
Your body already has T cells, white blood cells whose job is to identify and destroy threats. In cancer, these T cells often fail to recognise tumour cells as dangerous. CAR-T therapy solves that by genetically reprogramming your own T cells to see cancer and destroy it with lethal precision.
Scientists take T cells from your blood, send them to a laboratory, and genetically engineer them to carry a special protein on their surface, the CAR. This receptor acts like a GPS, locking onto specific proteins (called antigens) on the surface of cancer cells. Once these supercharged cells are grown into the hundreds of millions, they are infused back into the patient’s body, where they hunt, attack, and kill cancer cells.
As Dr Renier J. Brentjens of Roswell Park Comprehensive Cancer Centre beautifully put it: “We are giving patients a living drug.”
That phrase captures everything. Unlike a pill or a chemical drip, CAR-T cells are alive. They multiply inside the body, persist over time, and keep fighting.
A Brief but Remarkable History
The journey of CAR-T cell therapy from laboratory idea to approved medicine is one of the most compelling stories in modern medicine.
- 1980s-90s: Scientists first conceptualised engineering T cells with artificial receptors.
- 2011: A landmark moment, a patient with advanced leukaemia at the University of Pennsylvania received an early CAR-T infusion under the team led by Dr Carl June. Within a month, the cancer was gone.
- 2017: The U.S. Food and Drug Administration (FDA) approves the first-ever CAR-T cell therapy tisagenlecleucel (Kymriah) to treat children with acute lymphoblastic leukaemia (ALL).
- 2024 onwards: Seven FDA-approved CAR-T therapies are now available, with over 1,580 clinical trials registered globally as of April 2024.
Was that the first patient treated in 2011? As of 2024, they remain cancer-free after 12 years, a testament to this therapy’s extraordinary potential.
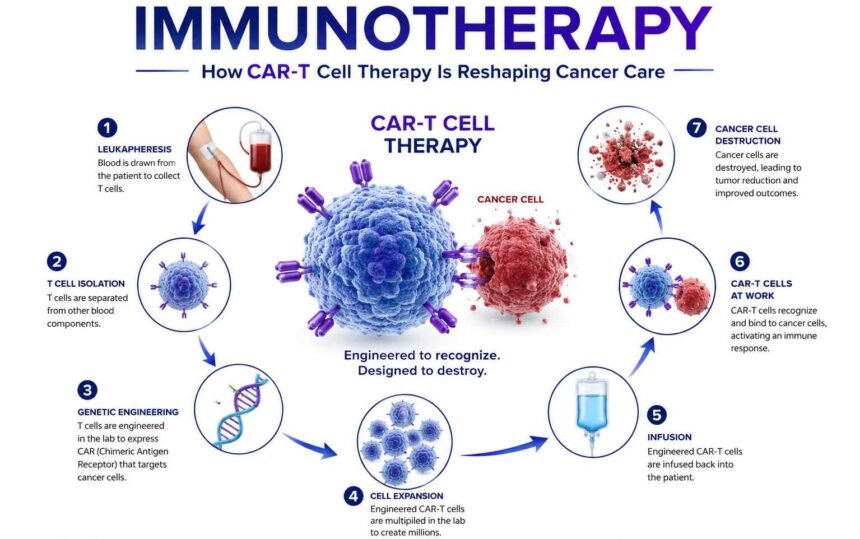
How Exactly Does It Work? The Step-by-Step Process
Understanding the process helps you appreciate just how personalised and sophisticated this treatment truly is.
| Step | What Happens |
| 1. Collection | Blood is drawn from the patient; T cells are separated out |
| 2. Engineering | T cells are sent to a lab and genetically modified to carry CARs |
| 3. Expansion | The modified cells are multiplied by hundreds of millions |
| 4. Infusion | The CAR-T cells are returned to the patient via a single IV infusion |
| 5. Activation | Inside the body, cells expand further and target cancer cells |
The entire process from blood draw to infusion currently takes 3 to 5 weeks. Each treatment is unique to the patient. It is, in every sense, personalised medicine.
What Cancers Can It Treat? Current Approvals and Results
CAR-T therapy has shown the most dramatic results in blood cancers, leukaemias, lymphomas, and multiple myeloma. The numbers speak for themselves:
- In a clinical trial involving patients with advanced follicular lymphoma, the CAR-T therapy eliminated cancer in nearly 80% of patients.
- In children with relapsed ALL (acute lymphoblastic leukaemia), tisagenlecleucel (Kymriah) produced remission in the majority of patients, many of whom appear to be cured.
- Depending on the patient’s characteristics and the type of blood cancer, response rates can vary from 20% to 90%.
FDA-Approved CAR-T Therapies (as of 2026)
| Therapy Name | Cancer Type |
| Tisagenlecleucel | B-cell ALL, Large B-cell Lymphoma |
| Axicabtagene ciloleucel | Large B-cell Lymphoma, Follicular Lymphoma |
| Brexucabtagene autoleucel | Mantle Cell Lymphoma, B-cell ALL |
| Lisocabtagene maraleucel | Large B-cell Lymphoma |
| Idecabtagene vicleucel | Multiple Myeloma |
| Ciltacabtagene autoleucel | Multiple Myeloma |
| Obecabtagene autoleucel | B-cell ALL |
In less than ten years, seven approved treatments have been developed; that is a revolution rather than a gradual improvement.
Challenge: Solid Tumours Are a Different Beast
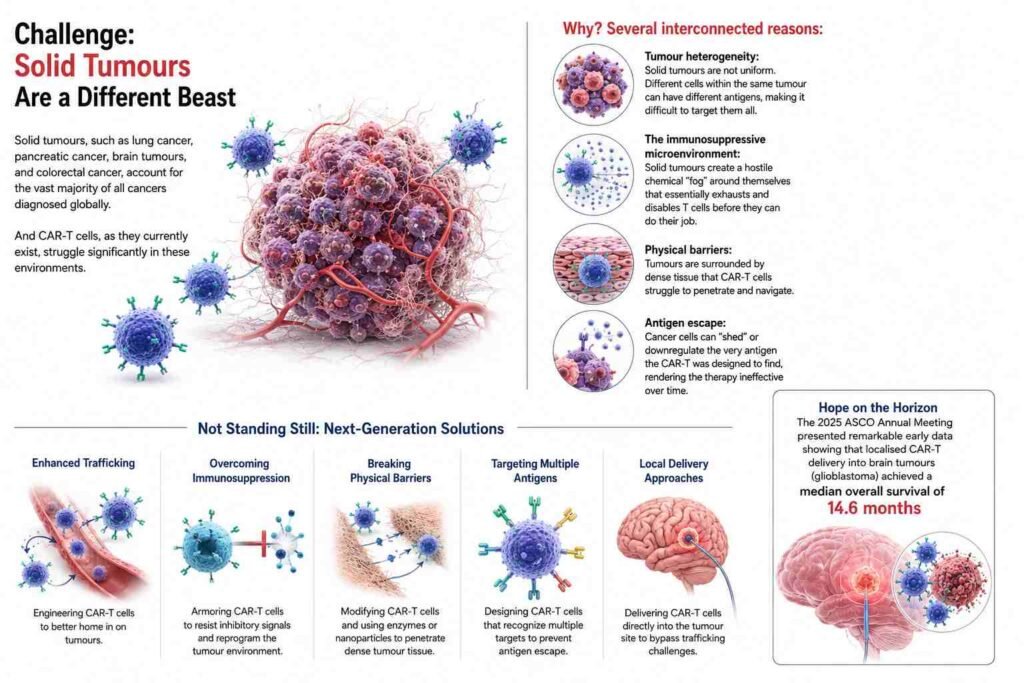
CAR-T therapy, for all its brilliance, has a major obstacle: solid tumours.
Solid tumours, such as lung cancer, pancreatic cancer, brain tumours, and colorectal cancer, account for the vast majority of all cancers diagnosed globally. And CAR-T cells, as they currently exist, struggle significantly in these environments.
Why? Several interconnected reasons:
- Tumour heterogeneity: Solid tumours are not uniform. Different cells within the same tumour can have different antigens, making it difficult to target them all.
- The immunosuppressive microenvironment: Solid tumours create a hostile chemical “fog” around themselves that essentially exhausts and disables T cells before they can do their job.
- Physical barriers: Tumours are surrounded by dense tissue that CAR-T cells struggle to penetrate and navigate.
- Antigen escape: Cancer cells can “shed” or downregulate the very antigen the CAR-T was designed to find, rendering the therapy ineffective over time.
However, the scientific community is not sitting still. Researchers are actively tackling these challenges through next-generation designs, and the 2025 ASCO Annual Meeting presented remarkable early data showing that localised CAR-T delivery into brain tumours (glioblastoma) achieved a median overall survival of 14.6 months in patients with recurrent disease, a cancer with notoriously grim outcomes.
What Are the Side Effects? Being Honest About the Risks
There are hazards associated with any effective treatment, and CAR-T therapy is no different. Patients and their families need to be educated because the adverse effects can be anything from tolerable to fatal.
The two most significant concerns are:
1. Cytokine Release Syndrome (CRS)
When CAR-T cells activate and multiply, they release large amounts of signalling chemicals called cytokines. In some patients, this becomes a runaway reaction, a “cytokine storm”, causing:
- Dangerously high fevers
- Severe drops in blood pressure
- In rare cases, it can be fatal
The good news: CRS can now be effectively managed with the drug tocilizumab, which targets the IL-6 receptor and has become the standard of care.
2. ICANS (Immune Effector Cell-Associated Neurotoxicity Syndrome)
Confusion, trouble speaking, and, in extreme situations, seizures are examples of neurological adverse effects. Steroids and supportive care are being used to treat these symptoms, and their use is being actively researched.
Other common side effects include:
- Infections (because the therapy can deplete B cells that produce antibodies)
- Persistent low blood cell counts (cytopenias)
- Fatigue and flu-like symptoms in the weeks following infusion
With improved patient monitoring systems, early interventions, and novel guidelines that improve safety results year after year, the field of managing CAR-T toxicity is developing at the same rate as the therapy itself.
Next Frontier: What’s Coming in CAR-T Science
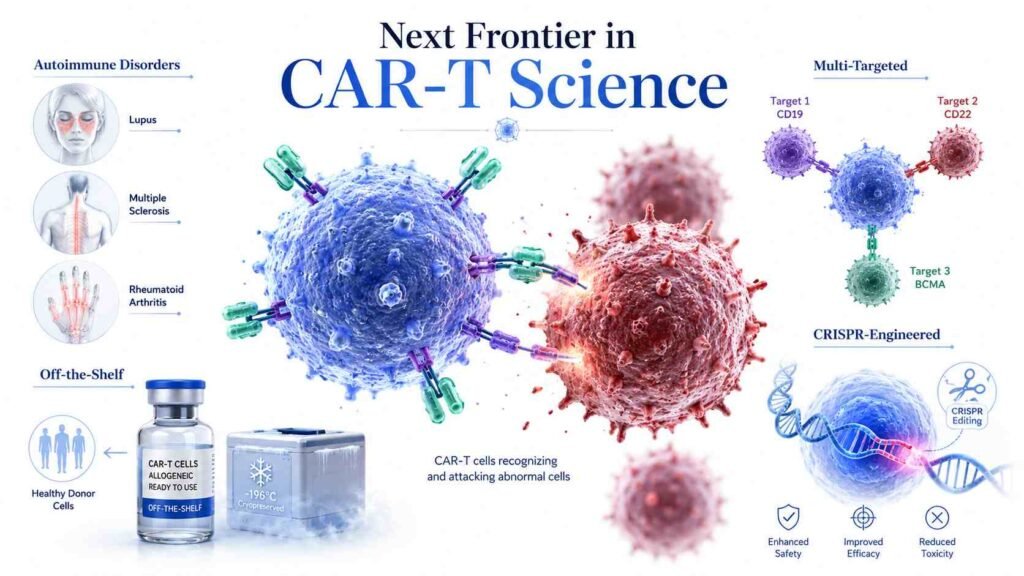
Most unexpectedly, scientists are currently investigating CAR-T therapy for autoimmune disorders such as rheumatoid arthritis, multiple sclerosis, and lupus. It might be used to eradicate the particular immune cells that trigger autoimmune attacks with the same accuracy that makes it effective against cancer cells. Early findings are encouraging and potentially change medicine in ways that go well beyond cancer.
“Off-the-Shelf” CAR-T Cells
Currently, every treatment is made from the patient’s own cells, a complex, expensive, and time-consuming process. The next revolution is allogeneic (donor-based) CAR-T cells, created from healthy donors and stored ready to use. These “off-the-shelf” therapies could:
- Dramatically reduce waiting time (from weeks to days)
- Lower costs significantly
- Make treatment accessible to patients who are too ill to wait
Multi-Targeted CAR-T Cells
Instead of targeting a single antigen, next-generation CAR-T designs target two or more antigens simultaneously. This reduces the risk of cancer “escaping” by shedding a single antigen, since the cells can still recognise the cancer via an alternative pathway.
CRISPR-Engineered CAR-T Cells
CRISPR gene-editing technology is being used to create more durable, more precise CAR-T cells, including ones that can resist the suppressive environment of solid tumours. Early studies have shown that TGF-β inhibition via CRISPR promotes long-term efficacy in solid tumour models.
CAR-T Beyond Cancer
Perhaps most surprisingly, researchers are now exploring CAR-T therapy for autoimmune diseases such as lupus, multiple sclerosis, and rheumatoid arthritis. The same precision that makes it powerful against cancer cells could be used to eliminate the specific immune cells that cause autoimmune attacks. Early results are promising, opening a door that could reshape medicine far beyond oncology.
Why This Matters: The Bigger Picture
For most of human history, cancer has been a sentence of suffering. The treatments we had were indiscriminate chemotherapy, which kills rapidly dividing cells, whether cancerous or healthy. Radiation damages tissue on both sides of the target.
CAR-T cell therapy is something fundamentally different. It is precision medicine in its most literal form, a treatment built from the patient’s own biology, engineered to target only the enemy, and capable of living on inside the body long after the infusion is complete.
The fact that the first patient treated over a decade ago is still cancer-free today is not just a statistic; it is proof of concept for a new chapter in medicine.
The road ahead still has real challenges: cost, accessibility, scalability, and the unsolved puzzle of solid tumours. But the direction is clear. CAR-T cell therapy is not a niche treatment for rare cases. As NCI pioneer Dr Steven Rosenberg said, “CAR T cells have become a part of modern medicine.”
Role of a CAR-T Specialist: Who Guides You Through This Journey?
Navigating CAR-T cell therapy is not something a patient does alone, and that’s where a CAR-T specialist becomes absolutely essential. A CAR-T specialist is a highly trained oncologist or haematologist with focused expertise in cellular immunotherapy, who oversees every stage of the treatment, from evaluating whether a patient is a suitable candidate to managing the complex process of cell collection, engineering coordination, and post-infusion monitoring.
These specialists work within dedicated academic medical centres and cancer institutes equipped to handle the unique demands of this therapy, including the rapid identification and management of side effects like CRS and ICANS. If you or a loved one is exploring CAR-T therapy as an option, seeking a consultation with a CAR-T specialist at a certified treatment centre is the critical first step, because in a therapy this precise therapy, the expertise guiding it matters just as much as the science behind it.
Choosing the Right CAR-T Cell Therapy Hospital
CAR-T cell therapy is performed in highly specialised cancer centres equipped with advanced immunotherapy infrastructure, expert oncology teams, ICU support, and specialized monitoring systems. Choosing the right CAR-T cell therapy hospital is critical because the treatment required close collaboration among haematologists, immunologists, laboratory experts, and rehabilitation professionals throughout the patient’s journey.
Many internationally recognized hospitals now provide full CAR-T programs that include personalized treatment planning, modern diagnostics, and long-term follow-up care for both domestic and overseas patients.
Final Conclusion
CAR-T cell therapy represents one of the most important advances in modern cancer treatment. Instead of relying only on external drugs, it uses the patient’s own immune system and gives T cells a new ability to recognise and attack cancer cells with greater precision. For several blood cancers, especially certain leukaemias, lymphomas, and multiple myeloma, CAR-T therapy has already changed outcomes for patients who previously had limited treatment options.
At the same time, CAR-T is still an evolving field. Challenges such as high cost, complex manufacturing, serious side effects, limited access, and weaker results in solid tumours remain important barriers. The next generation of CAR-T science is focused on solving these problems through off-the-shelf cell products, multi-targeted designs, CRISPR-engineered cells, and new applications beyond cancer, including autoimmune disease.
The future of CAR-T therapy is not just about treating more cancers. It may also reshape how doctors think about immune-based diseases as a whole. While more research is needed, the progress so far shows that living cell therapies are becoming a serious part of modern medicine, not just an experimental idea.
Medical Disclaimer
This article is for educational and informational purposes only. It should not be used as medical advice, diagnosis, or treatment guidance. CAR-T cell therapy is a highly specialised treatment that may involve serious risks, including cytokine release syndrome, neurological side effects, infections, and prolonged blood cell problems. Patients should always speak with a qualified oncologist, haematologist, immunologist, or certified CAR-T treatment centre before making any treatment decision.
Treatment availability, eligibility, safety, and outcomes can vary depending on the patient’s diagnosis, disease stage, previous treatments, overall health, country, hospital, and current clinical guidelines.
References
- June, C. H., & Sadelain, M. “Chimeric Antigen Receptor Therapy.” New England Journal of Medicine, 2018; 379(1): 64–73. DOI: 10.1056/NEJMra1706169.
- Porter, D. L., Levine, B. L., Kalos, M., Bagg, A., & June, C. H. “Chimeric Antigen Receptor-Modified T Cells in Chronic Lymphoid Leukemia.” New England Journal of Medicine, 2011; 365(8): 725–733. DOI: 10.1056/NEJMoa1103849.
- Maude, S. L., Laetsch, T. W., Buechner, J., et al. “Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia.” New England Journal of Medicine, 2018; 378(5): 439–448. DOI: 10.1056/NEJMoa1709866.
- Neelapu, S. S., Locke, F. L., Bartlett, N. L., et al. “Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma.” New England Journal of Medicine, 2017; 377(26): 2531–2544. DOI: 10.1056/NEJMoa1707447.
- Schuster, S. J., Bishop, M. R., Tam, C. S., et al. “Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma.” New England Journal of Medicine, 2019; 380(1): 45–56. DOI: 10.1056/NEJMoa1804980.
- Park, J. H., Rivière, I., Gonen, M., et al. “Long-Term Follow-up of CD19 CAR Therapy in Acute Lymphoblastic Leukemia.” New England Journal of Medicine, 2018; 378(5): 449–459. DOI: 10.1056/NEJMoa1709919.
- Lee, D. W., Santomasso, B. D., Locke, F. L., et al. “ASTCT Consensus Grading for Cytokine Release Syndrome and Neurologic Toxicity Associated with Immune Effector Cells.” Biology of Blood and Marrow Transplantation, 2019; 25(4): 625–638. DOI: 10.1016/j.bbmt.2018.12.758.
- Majzner, R. G., & Mackall, C. L. “Clinical Lessons Learned from the First Leg of the CAR T Cell Journey.” Nature Medicine, 2019; 25: 1341–1355. DOI: 10.1038/s41591-019-0564-6.
- Depil, S., Duchateau, P., Grupp, S. A., Mufti, G., & Poirot, L. “‘Off-the-Shelf’ Allogeneic CAR T Cells: Development and Challenges.” Nature Reviews Drug Discovery, 2020; 19: 185–199. DOI: 10.1038/s41573-019-0051-2.
- Mackensen, A., Müller, F., Mougiakakos, D., et al. “Anti-CD19 CAR T Cell Therapy for Refractory Systemic Lupus Erythematosus.” Nature Medicine, 2022; 28: 2124–2132. DOI: 10.1038/s41591-022-02017-5.
- Melenhorst, J. J., Chen, G. M., Wang, M., et al. “Decade-Long Leukaemia Remissions with Persistence of CD4+ CAR T Cells.” Nature, 2022; 602: 503–509. DOI: 10.1038/s41586-021-04390-6.